Dr.-Ing Martin Sörenson
CEO, enviolet Corp.
Dipl. Ing. Jürgen Weckenmann
Technical Director, enviolet Corp.
Dipl. Ing. Frank Zegenhagen
Senior R&D Manager, enviolet Corp.
Triton X-100 is a non-ionic surfactant with numerous applications in the life science and pharmaceutical industries, for example as a general-purpose detergent or as a viral inactivation agent in medicinal products. However, this widely used chemical – whose degradation byproducts have endocrine disrupting properties with probable serious effects on many organisms, including humans [1] – is also a cause for concern and, thus, its release into the aquatic environment via industrial wastewaters should be prevented. For this reason, it has been placed onto Annex XIV of the REACH Regulation, which aims to protect human health and the environment from possible risks posed by chemicals.
Biological wastewater treatment is not suitable in industrial applications, since typical concentrations of Triton X-100 far exceed the tolerance of aerobic and anaerobic organisms capable of biodegrading this substance [2]. Additionally, long treatment time – due to organism acclimation – further reduces the applicability of biological treatment systems [3]. The current standard procedure at many industrial facilities is to collect the contaminated wastewaters on site for transfer to off-site thermal oxidation, a very cost- and energy-intensive method in which polluted wastewaters are essentially incinerated. However, UV oxidation presents a more efficient and extensively proven method for the destruction of Triton X-100 in industrial wastewaters.
What is UV oxidation?
UV oxidation is a chemical photo-oxidation process in which UV light is absorbed by a molecule to produce excited electrons. For example, UV irradiation can split hydrogen peroxide into highly reactive hydroxyl radicals that react quickly with organic and inorganic compounds in aqueous solutions. Depending on the composition of a wastewater, different reaction pathways are initiated by these free radicals. With proper process control, these reaction pathways can be exploited to meet various degradation goals, i.e. selective oxidation of single compounds or complete mineralization with H2O and CO2 as reaction products of the original organic compounds. The intermediates – which contain one or several -OH, =O and COOH groups – are typically considerably less toxic than the original compounds and exhibit increased bioavailability characteristics.
The wavelength used in this UV application also leads to direct photolytic processes, generating radicals within the octylphenols, a class of compounds cumulatively referred to as Triton X, to increase the overall reaction efficiency.
Feasibility study
The first step in any UV oxidation plant design is a laboratory evaluation into the feasibility with regards to the reduction targets. For wastewaters containing Triton X-100 there are three main objectives:
- Destruction of Triton X compounds
- Reduction of chemical oxygen demand (COD), which is a measure quantifying the amount of oxidizable pollutants in the water
- Increase of biological oxygen demand (BOD) and the biodegradability of the organic compounds remaining in the effluent after treatment
Since the treatment class, a measure of how easily a substance can be oxidized, for Triton X-100 is already known, a preliminary microtest can be forgone. The macrotests – performed as a batch treatment with a 40W laboratory-scale UV reactor – then are carried out to determine the proper UV dosage required in order to achieve the objectives. The batch process means that the sample is circulated through the laboratory reactor, which is functionally equivalent to an industrial scale plant, while H2O2 dosing is monitored and controlled.
At several intervals during the treatment, samples are extracted in order to analyze various parameters. Total organic carbon (TOC), COD and BOD5 are measured in house. A rough indication of the Triton X-100 level can be gained using a diode array detector (DAD), a cost-effective method of monitoring the destruction of Triton X-100. More precise external analytics are performed for an initial validation of the correlation results. Samples also can be returned to the customer for independent testing.
Results
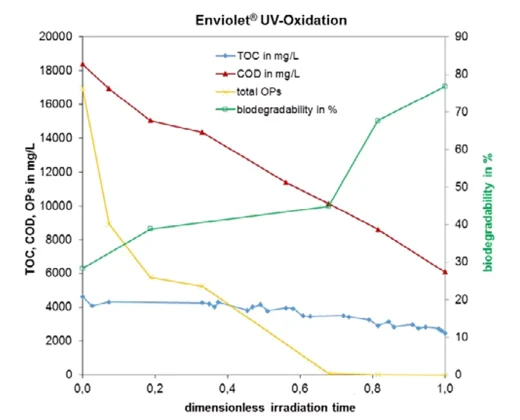
Original samples contained elevated levels of Triton X-100, as well as several other octylphenol compounds. Figure 1 shows the reduction in the sum of all octylphenols (including Triton X-100), as well as a reduction in the COD and an increase in the biodegradability of one set of samples.
The oxidation of initial structures results in several degradation pathways leading to the formation of smaller molecules, mainly carboxylic acids. As the organic acids are degraded, the TOC decreases as a result of the organic carbon being further oxidized to carbon dioxide.
These reaction pathways are reflected in the change in the COD/TOC ratio, from around three to four in the beginning of the reaction to lower ratios during treatment, as seen in Figure 1 [4]. With increasing treatment time, less oxygen (COD) is necessary to oxidize the remaining TOC, as this carbon is extensively oxidized by the progression of the treatment process.
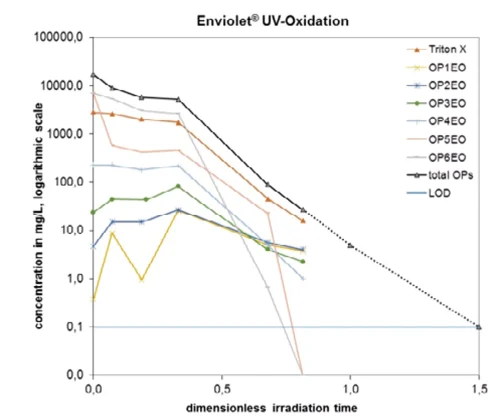
Figure 2 illustrates the degradation of Triton X-100 and the other examined octylphenols (OP1EO = octylphenol monoethoxylate – through to OP6EO = octylphenol hexaethoxylate. Initially, the levels of some of the octylphenols with shorter ethoxylate chains remain constant or even rise since they are formed as intermediate degradation products but are later lowered significantly as the reaction time increases.
During laboratory simulation, the normalized dimensionless treatment time is used because actual treatment hours can only be determined after final levels of octylphenol compounds are determined and all design information for the commercial plant is known, e.g. flow rates, charge volume and power requirements.
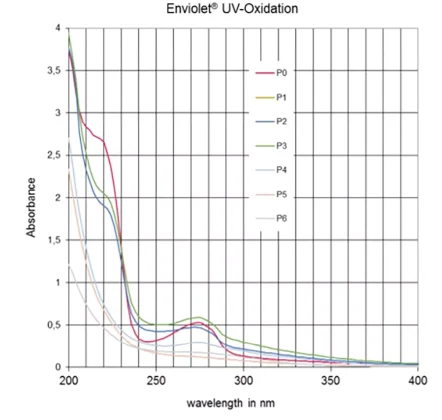
The laboratory simulation ends when the DAD test produces no absorption peak in the 260 to 290 nm wavelength range [5], which is characteristic of solutions containing Triton X-100 (Figure 3).
Industrial implementation
A resulting full-scale industrial plant can be designed for batch treatment in which wastewaters are collected in a large storage tank from various production lines or for several days, depending on the wastewater flow rate. Before being pumped in a loop through the UV reactors, the wastewaters are dosed and thoroughly mixed with H2O2, a safe, stable and low-cost oxidizing agent. A special reactor design creating high turbulence ensures an optimal UV treatment, even with highly absorbing or turbid media.
This batch treatment process has the advantage of being highly flexible with regards to varying wastewater flows, as well as to fluctuating concentrations of the targeted compounds. The standard operating procedure (SOP) ensures simplified handling and allows the user to independently adapt the treatment time and parameters to a fluctuating wastewater matrix.
A turnkey enviolet oxidation system includes a batch tank, dosing stations for the required chemicals, static mixer, media pump, enviolet® UV reactors, all equipment for continuous and automated monitoring of process parameters (pH, temperature etc.), as well as piping and a heat exchanger. The modular design allows for easy up-scaling, and the footprint of these compact installations is modest and can often fit neatly into preexisting structures, e.g. wastewater collection tanks. Customer-specific space constraints can also be addressed during the planning phase. Installation solutions can be internal, external or a combination approach.
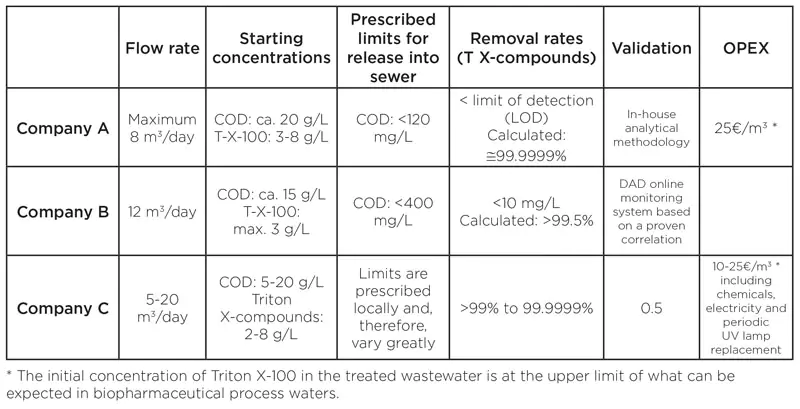
Table 1 shows typical data from Triton X applications. Depending on the initial concentrations of Triton X-100 and the COD levels, as well the possible presence of further targeted substances, the operating costs – which include all consumables, electrical energy, spare parts and maintenance – are between 10 to 25 Euros ($11.20 to $28) per cubic meter. This is a considerable costs savings compared to other conventional disposal methods and, in most cases, the initial capital expenditure is amortized within a short period of time [6].
The reduction of all Triton X compounds to below the limit of detection ensures that uncertainty regarding the development of binding EU guidelines is eliminated and that the plant can run without modification, regardless of where the limits may lie in the future. The same applies to the reduction of COD and the increase of biodegradability. Both measures achieve levels that far exceed what is considered standard good practice.
Summary
This application exemplifies the efficacy of advanced UV oxidation of industrial wastewater containing xenobiotic substances. Triton-type molecules can be easily and reliably removed by this technique, even in complex wastewater matrixes. A specially developed analytical monitoring method facilitates automation and reduces the labor required for operation. The low operating expense in relation to other treatment methods leads to considerable cost savings. Although there also are systems treating Triton in sewage treatment plants, the elimination rate of targeted molecules is not high enough to justify the required effort, in contrast to cases in which target molecules are treated close to the source.
In general, the UV oxidation method is highly flexible and can be adapted to target a host of other compounds, such as active pharmaceutical ingredients (APIs) or toxic manufacturing by-products. A comprehensive laboratory simulation can pinpoint the best oxidation procedure for eliminating undesired compounds according to different goals, i.e. release to municipal wastewater treatment plant or even directly into the environment. The high efficacy, even reduction below the detection limit, provides long-term planning certainty in the face of ever stricter wastewater regulations. n
enviolet GmbH has been installing tailor-made industrial AOP plants to service specific client requirements since 1997 and has more than 600 AOP plants in operation worldwide. enviolet’s core industrial applications are used for heavy-duty industrial applications in pharmaceutical, surface, chemical, aircraft and automotive industries, as wall as waste disposal. For more information, visit www.enviolet.com.
References
[1] D. T. S. H. H.J. Chen, „Biodegradation of octylphenol polyethoxylate surfactant TritonX-100 by selected microorganisms.,“ J. Bioresource Technol., pp. 1483-1491, 2005.
[2] M. B. M. F. Lina Abu-Ghunmi, “Fate of Triton X-100 Applications on Water and Soil,” Journal of Surfactants and Detergents, pp. 833-838, September, 17(5) 2014.
[3] M. K. M.-J. A. K. J. Liwarska-Bizukojc E, „Acute toxicity and genotoxicity of five selected anionic and nonionic surfactants,“ Chemosphere, pp. 1249-1253, March, 58(9) 2005.
[4] F. Zegenhagen, „Feasibility Study: UV-Oxidation of Organic Matter (COD) in a pharmaceutical waste water,“ Karlsruhe, Germany, 2018.
[5] Merck KGaA, Darmstadt, Sigma Product Information Sheet, Triton X-100™.
[6] S. e. al., „Wastewater Treatment by UV-Oxidation at Oril API Manufacturing Site,“ Pharm. Ind., pp. 569-576, 80(4) 2018.