By Mike Reeves, EIT2.0
UV monitoring of HVAC systems in pharmaceutical facilities is essential to maintain contamination control, validate germicidal performance over time and satisfy growing expectations from regulators and quality auditors that air treatment processes be measurable, documented and continuously verified. 1, 2, 3 Without direct monitoring of UV dose in ducts, air handlers and coils, pharma manufacturers risk drift in disinfection performance as lamps age, fouling accumulates and operating conditions change – creating hidden vulnerabilities in cleanroom and support-area biosecurity.4, 5, 6
Role of HVAC in Pharma Contamination Control
Pharmaceutical HVAC systems act as the backbone of environmental control, governing temperature, humidity, airflow direction and particle counts in production and support spaces. 1, 7 Cleanrooms, weighing areas, formulation suites and packaging zones depend on tightly controlled, filtered air to keep viable and nonviable contamination within specified limits for each room classification. 1, 8
- HVAC designs for pharma incorporate HEPA/ULPA filtration, pressure cascades and defined air change rates to ensure unidirectional flow over critical processes and to prevent cross-contamination between rooms of different cleanliness grades. 1, 7
- As more facilities add UV-C to ducts, coils or upper-air zones as a supplementary biocidal layer, the HVAC system becomes not only a carrier of clean air but also a primary platform for continuous microbial risk reduction. 4, 5, 6
Because so much of product and patient safety depends on HVAC performance, any added germicidal technology in that path must be monitored with the same rigor as temperature, humidity and pressure.
Why UV is Used in Pharma HVAC
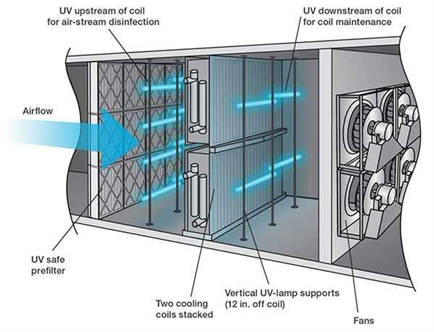
UV-C, typically in the germicidal range around 254 nm (and increasingly in UV-C LED bands), is used to inactivate microorganisms by damaging their nucleic acids so they cannot replicate. 5,3 When integrated into HVAC, these systems target both airborne microorganisms in the airstream and biofilm or microbial reservoirs on coils, drain pans and duct surfaces. 4, 9, 5
- Coil/surface irradiation systems continuously operate to prevent mold, bacteria and biofilm from establishing on cooling coils and condensate pans, which otherwise can shed viable organisms into critical airstreams or degrade heat-transfer efficiency. 9, 5
- Airstream disinfection systems are placed in plenums or ducts to deliver a calculated UV dose based on intensity, exposure time and target organism susceptibility, reducing the concentration of airborne pathogens “on the fly.” 4, 5, 6
Studies and guidance documents show that properly designed UV-C systems can achieve up to 99% inactivation of microorganisms on HVAC surfaces and substantial reductions in airborne pathogen levels, enhancing indoor air quality and lowering infection risks. 5, 3 For pharmaceutical manufacturers, this supports both product protection and the protection of immunocompromised patients in hospital-based or clinical manufacturing settings.
Results of Unmonitored UV in HVAC
Despite these benefits, UV systems are not “set and forget.” Without monitoring, their actual delivered dose can deviate significantly from the design assumptions over time, undermining the intended level of protection. 4, 5, 6 Several factors drive this drift:
- Lamp aging and output decay: Low-pressure mercury and other UV lamps gradually lose output over their service life, often dropping 20-40% or more before end-of-life, which directly reduces germicidal dose if power is not adjusted or lamps are not replaced on the correct schedule. 5, 3
- Fouling and optical obstruction: Dust, film, condensate or chemical deposits on lamps, sleeves and surrounding surfaces can attenuate UV intensity, especially in humid or coil-proximate locations where fouling is common. 4, 9
- Changes in airflow and temperature: Variations in air velocity, temperature and humidity in the duct or plenum alter pathogen residence time and can affect lamp output, changing the effective dose compared with initial design conditions. 1, 5
If these effects are not measured, UV systems may remain powered and “on” while delivering a sub-therapeutic dose that no longer meets the assumed log reduction for key organisms. For a pharmaceutical plant relying on these systems to control airborne bioburden, this creates a silent gap in the contamination control strategy that may only be detected during a deviation, excursion or adverse audit finding.
Regulatory and Quality Expectations in Pharma
While major regulations (e.g., FDA cGMP, EU GMP Annex 1, USP chapters such as 797/800) focus more explicitly on particulate control, pressure differentials and temperature/humidity, they consistently emphasize documented control of contamination risks and verification of critical environmental systems. 1, 8, 7 As UV becomes part of the contamination control strategy, regulators increasingly expect it to be treated as a critical utility or critical control parameter rather than a nonqualified accessory.
- Guidance on sterile compounding and hazardous drug handling (for example, USP 797 and 800) highlights the necessity of continuous monitoring of environmental parameters that impact sterility and product integrity, such as airflow, pressure and temperature; when UV is introduced as a biocidal mechanism, it logically falls into the set of systems whose performance must be demonstrated and maintained. 1, 8
- FDA expectations for facilities and equipment under cGMP include evidence that environmental controls are designed, qualified and operated within defined limits, with appropriate alarms, documentation and maintenance; a UV-disinfecting HVAC section used to support aseptic operations would be scrutinized under the same lens. 7, 8
Quality management systems built around risk-based contamination control strategies (as seen in Annex 1 revisions and industry best practices) encourage manufacturers to identify critical steps in air and surface decontamination and implement monitoring commensurate with their risk impact. For UV HVAC systems, this increasingly points toward real-time or at least periodic measurement of actual UV performance, not only indirect proxies, such as lamp hours.
How UV Monitoring Enhances Risk Management

Dedicated UV sensors and monitoring systems integrated into HVAC UV installations provide objective data on lamp output, dose and system status, which can be used to improve risk management across the life cycle of the installation. 4, 5, 6 This aligns UV systems with the same data-driven philosophy used for other critical utilities.
Key Risk Management Benefits Include:
- Verification of delivered dose: Online UV sensors can measure intensity at critical locations, allowing calculation or confirmation of delivered dose in coils or airstreams and ensuring it remains above the minimum required for target organisms. 4, 5
- Early detection of performance drift: Trending UV intensity over time helps detect gradual lamp degradation, fouling or system imbalance before the germicidal margin of safety is eroded, enabling predictive maintenance rather than reactive corrections after a deviation. 4, 6
- Alarm and interlock functionality: Continuous monitoring allows integration with building management systems so that low-UV conditions trigger alarms, fault logs, or even interlocks where necessary for high-risk zones, similar to how pressure and temperature excursions are handled. 4, 1
From a pharmaceutical quality risk management perspective, these capabilities reduce the probability of undetected loss of disinfection performance and provide documented evidence that the UV element of the HVAC system is functioning as intended.
Support for Validation and Qualification
When UV systems are monitored, they become significantly easier to validate, requalify and defend during audits because performance can be linked directly to measured, recorded parameters. 4, 5, 3 Validation teams can build protocols that tie together dose targets, sensor readings and microbiological outcomes.
- During initial qualification (IQ/OQ/PQ), UV monitoring data can be collected while performing microbiological challenge studies or environmental monitoring to correlate measured dose to achieved log reduction or bioburden trends in critical areas. 5, 3
- Once correlated, routine monitoring limits and alarm thresholds can be established; periodic requalification then focuses on confirming that the relationship between sensor readings and microbial control outcomes remains valid as lamps and equipment age. 4, 3
This approach mirrors how other critical utilities (e.g., purified water systems, clean steam) are managed: measurable critical quality attributes and critical process parameters are identified, controlled and trended, and deviations prompt investigations and CAPAs. Without UV monitoring, validation efforts are forced to lean heavily on indirect measures, such as lamp specifications and replacement intervals, which are harder to defend under modern risk-based regulatory scrutiny.
Integration with Building Automation and Data Systems

Modern UV monitoring hardware can integrate with building automation systems (BAS) or facility management platforms, where pharmaceutical manufacturers already supervise HVAC parameters including temperature, humidity, pressure and airflow. 4, 1 Consolidating UV performance data with these other variables creates a more complete picture of environmental control and simplifies both operations and documentation.
- Real-time dashboards can display UV intensity or “lamp health” alongside HEPA filter status, air change rates and room classifications, making it easier for operators and engineering teams to spot correlations and anomalies. 4, 1
- Historical data logging supports trending analyses required by internal quality review, management review and regulatory inspections and can feed into continuous improvement programs and energy optimization efforts. 4, 3
For multi-site pharmaceutical organizations, standardized UV monitoring across facilities also enables benchmarking of performance, harmonization of maintenance strategies and centralized oversight of contamination control technologies.
Operational and Economic Benefits
Beyond regulatory alignment and risk reduction, UV monitoring of HVAC in pharmaceutical environments delivers tangible operational and cost benefits when correctly implemented. 4, 9, 5 These gains support business cases for investment in both UV and monitoring hardware.
- Optimized lamp replacement: Rather than replacing lamps strictly on worst-case time-based schedules, monitored systems can use actual output data to determine replacement timing, balancing assurance of disinfection with reduced consumable costs. 4, 5
- Improved coil performance and energy efficiency: By ensuring UV output remains sufficient to prevent biofilm accumulation on coils, monitored systems help maintain design heat transfer efficiency, reduce fan energy (through lower pressure drops) and avoid costly coil cleaning or replacement. 9, 5
- Reduced downtime and contamination-related losses: Faster detection of underperforming UV systems reduces the likelihood of environmental excursions, batch rejections or production shutdowns associated with contamination events, which can exceed the cost of monitoring infrastructure. 5, 3
In an industry where unplanned downtime, product loss and regulatory remediation can each carry high six- or seven-figure costs per incident, modest investments in UV monitoring can deliver an attractive risk-adjusted return.
Alignment with Post-COVID Expectations and Future Trends
Post-COVID-19, expectations for air quality and airborne infection control have increased across regulated and healthcare-related environments, including pharmaceutical manufacturing and associated laboratories. 2,3,6 UV-C in HVAC now is recognized not just as an energy- or maintenance-focused technology, but as part of a broader infection prevention and contamination control strategy that must be demonstrable and auditable.
- Industry analyses emphasize that UV-C can continuously reduce microbial loads in HVAC systems and indoor air. Still, analyses stress that system design, dose and maintenance are critical to achieving the promised risk reductions. 5, 3
- As pharma organizations adopt more advanced digitalization and Industry 4.0 concepts, including real-time environmental monitoring and advanced analytics, UV monitoring becomes a natural extension, supplying a previously missing data stream about an important biocidal mechanism. 4, 1
Emerging technologies such as Far UV-C, UV-C LEDs and hybrid air treatment systems will further expand UV’s role in HVAC. For each of these technologies, robust monitoring will be key to demonstrating consistent performance, building internal and regulatory confidence and supporting the evolution of standards and best practices in pharmaceutical environmental control. 5, 6
In sum, UV monitoring of HVAC in the pharmaceutical industry is needed because UV has moved from a peripheral add-on to a critical component of contamination control, and critical components must be measured, trended and controlled – not assumed. Continuous or periodic measurement of UV performance closes a major visibility gap in air and surface disinfection, strengthens validation, supports compliance and reduces both operational and patient safety risks across the facility lifecycle. 4, 1, 5
References
- Air Innovations. Pharmaceutical Temperature and Humidity Control Solutions. Air Innovations. https://airinnovations.com/industries/life-sciences-pharmaceutical-hvac/
- Bruno, L. (n.d.). Operating a pharmaceutical HVAC system in a post COVID-19 environment. IPS-Integrated Project Services, LLC. https://www.ipsdb.com/happenings/insights/operating-a-pharmaceutical-hvac-system-in-a-post-covid-19-environment
- American Society of Heating, Refrigerating and Air-Conditioning Engineers. (2019). Ultraviolet air and surface treatment. In 2019 ASHRAE handbook: HVAC applications (Chapter 62). https://www.ashrae.org/file%20library/technical%20resources/covid-19/i-p_a19_ch62_uvairandsurfacetreatment.pd
- Ultravation, Inc. Commercial indoor air quality products. Ultravation Commercial. https://ultravationcommercial.com/ultravation-commercial-indoor-air-quality-products/commercial-contact/
- Skelton, D. (2020). UV-C for HVAC air and surface disinfection. AMCA inmotion. Air Movement and Control Association International. https://www.amca.org/educate/articles-and-technical-papers/amca-inmotion-articles/uv-c-for-hvac-air-and-surface-disinfection-2.html
- Hughes Environmental. (n.d.). UVGI light disinfection: A guide for healthcare facilities. https://hughesenv.com/uvc-light-disinfection/
- U.S. Food and Drug Administration. (2015, July 14). Facilities and equipment: CGMP requirements [Presentation]. U.S. Department of Health and Human Services. https://www.fda.gov/downloads/drugs/developmentapprovalprocess/smallbusinessassistance/ucm456370.pdf
- QUALIA. (2024, December 22). FDA regulations for air showers: Compliance. QUALIA Bio. https://qualia-bio.com/blog/fda-regulations-for-air-showers-compliance/
- American Ultraviolet. (n.d.). HVAC on-coil (UV germicidal solutions). https://www.americanultraviolet.com/uv-germicidal-solutions/hvac-on-coil.cfml
- Nuvonic. (n.d.). UV water treatment for pharmaceutical industries (Pharmaceuticals & healthcare). https://www.nuvonic.com/industries/pharmaceuticals-healthcare/
Mike Reeves brings more than 30 years of experience in the medical device industry, combining technological expertise with a deep passion for improving healthcare outcomes. Throughout his career, he has earned recognition as a thought leader for his ability to anticipate market needs, identify emerging trends and drive innovation that enhances patient care. His proven track record spans product development, regulatory compliance and business growth, supported by a commitment to maintaining the highest standards of quality and safety. Today, Reeves shares EIT2.0’s mission to make healthcare environments safer through UV measurement and disinfection technology. His dedication, expertise and drive to create meaningful change position him to make a lasting impact on both the future of medical technology and the quality of patient care. For more information, visit www.eit20.com.

