By Arthur Kreitenberg M.D. FACS, Clinical Professor of Orthopedic Surgery, University of California Irvine, and James P. Malley, Jr, Ph.D., Editor-in-Chief, UV Solutions and Emeritus Professor of Civil and Environmental Engineering, University of New Hampshire
“Hygiene Theater” 1 for the purpose of this article is defined as the practice of visible but ineffective cleaning rituals based on surface-level optics and not on scientifically backed measures that address how infections in healthcare facilities are spread. The COVID-19 pandemic and healthcare acquired infection (HAI) concerns fueled widespread fundamental and applied research and testing of commercially available approaches and devices aimed at improving indoor air quality, targeted cleaning of high-touch surfaces (HTS), and verification through testing/monitoring and effective communication of the infection control measures being implemented.
A basic UV-C technology development roadmap to achieve true improvement in healthcare facility hygiene is presented, which: a) begins with understanding the overall disease transmission pathway for a specific facility and the role a given UV-C application is attempting to fill; b) employs UV-C technology that has been validated for the specific full-scale application in which it will be used; and c) essential monitoring to ensure the UV-C technology is operating according to specifications and signaling when maintenance is needed. The article will conclude with a “first do no harm” discussion based on practices involving UV-C technologies that should not occur.
The key takeaway of this paper is that for the benefit of public health, as well as the advancement of the ultraviolet technology industry, it is imperative that professional associations, through their publications, report on emerging UV-C technologies and practices for infection control; emphasize those that work in practical, full-scale, applications; and clearly identify those whose value is dubious or requires further research, development and successful full-scale demonstration.
Understanding Facility-Specific Modes of Disease Transmission
The chain of infection often is broken into six steps: infectious agent, reservoir, portal of exit, mode of transmission, portal of entry and susceptible host. 2 Most UV-C devices are developed to degrade or break the chain at the mode of transmission. When a UV-C device is considered for a healthcare application, two types of transmission often are the focus: high-touch surfaces and indoor air quality (specifically aerosol transmission). For demonstrative purposes, two applications will be considered: UV-C on high-touch surfaces (HTS) and upper room UV germicidal irradiation (URUVGI).
High-Touch Surfaces (HTS)
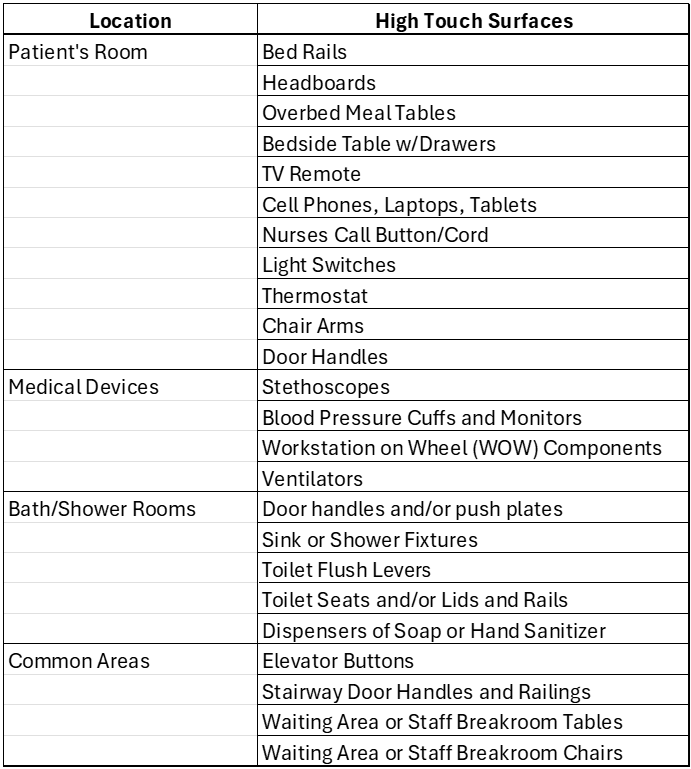
In a typical healthcare setting, studies to reduce HAI identified many common HTS, as shown in Table 1. 4 Recent innovations in infection control have introduced automated UV-C-based enclosures for HTS areas, such as elevator controls, bathroom fixtures, handrails, etc.
Theoretical Efficacy vs. Real-World Limitations
The engineering specifications of available products, when supported by validation testing, can provide a rapid, significant log-reduction of relevant viral and HAI pathogens, as well as user protection from stray UV-C exposures that could exceed OSHA guidelines. 3 However, their efficacy as an infection control approach needs to be measured by impact on the entire chain of transmission. The practical impact of these localized and specialized approaches on pathogen transmission or disease prevention is not well documented in the reviewed literature.
The Problem of Selective Protection: Site-specific UV-C “enclosure devices” often are intended for building systems that consist of multiple touchpoints. For example, effective disinfection of the interior elevator buttons while ignoring the hallway “call” buttons cannot effectively break this potential chain of transmission. Similarly, site-specific UV-C “enclosure devices,” which can effectively disinfect bathroom stall door latch points, do not impact other bathroom HTS (see Table 1).
Secondary Surface Contamination: Pathogen transfer is not limited to the elevator interior buttons. Users frequently contact elevator railings, walls and mirrors. Users of the toilet likely are to touch the toilet seat, flush lever, toilet paper dispenser and handicap rails. A localized system may fail to address all the high-touch environment points, even in simple applications, rendering overall infection control inadequate.
Statistical Impact: Because human movement involves contact with tens to hundreds of HTS daily, the isolation of specific surfaces (see Table 1) has not been demonstrated in published literature to yield a statistically significant reduction in individual or public health infection rates in healthcare facilities.
Upper Room UV Germicidal Irradiation (URUVGI)
Upper room UVGI (URUVGI) effectiveness often is cited in literature as highly effective for tuberculosis and measles transmission, with a large body of promising results published between 1930 and 1950. 5, 6, 7 Careful review of those early studies, as well as studies intended to replicate their findings, highlights vital details that can be overlooked easily.
The first involves defining what is meant by “promising.” Modern UV-C disinfection claims and goals, for a myriad of reasons, have been influenced heavily by competitive product marketing vs. chemical disinfectants and define effectiveness as 99% to 99.99% or higher inactivation of target microbes. This stands in sharp contrast to detailed studies reported in the literature, where the effectiveness of URUVGI often is linked to reductions in the “microbial load” measure of about 50% to 80%. 5, 6, 7 Similarly, the literature often cited for URUVGI’s ability to combat measles is based on work with schools in the 1930s, when the infection rates between UVGI-treated classrooms and non-treated classrooms reduced the measles transmission by about 30%. 7
In addition, careful study of the URUVGI system effectiveness highlights the importance of understanding UV-C fixture efficiencies in terms of actual delivered UV-C, air mixing within the treated space and overall incidence of infection rates in the population outside of the treated space. 5, 6, 7 The latter is particularly critical to interpreting results. For example, when URUVGI is applied in a controlled healthcare (or school classroom) environment, but the external environment experiences a high incidence of that disease, such as during a measles outbreak, then the prior results demonstrating the effectiveness and benefit of URUVGI cannot be reproduced. 5, 6, 7
The efficacy and limitations discussed previously also apply to URUVGI applications. Consider a recent trial examined by researchers at University of New Hampshire (UNH) of a 100-bed skilled nursing facility (aka sniff or nursing home) in New England that conducted its own on-site experiment by placing URUVGI devices in common dining/activity rooms. The basis for equipment selection, the number and the placement of devices was undocumented and left to the discretion of the equipment sales staff. In addition, no measurements (e.g., see reference 5 of Visual-UV site-specific calibrated software demonstrating an average room fluence rate of 5 to 7 μW/cm2) were made during the trial period. The conclusion from the UNH peer review of these results, conducted after the fact, was that no statistically significant reduction in cold and flu cases among residents during the period December 2024 through February 2025 was achieved when compared to the prior five years.
In the context of this paper’s recommendations, the results would not be surprising, since the disease transmission pathways for this facility were not clearly understood, given that most residents did not use common dining areas but chose to dine in their own rooms. Communal areas that were used frequently by the residents, including hallways, a central recreation room and shared restroom/bathing facilities, did not receive URUVGI or HTS UV treatment. In addition, the URUVGI installation was neither validated nor monitored for achieving a specified fluence rate in the communal areas treated.
UV Equipment/Facility Validation

From 2020 to the present, UNH has been involved in the laboratory evaluation of 30 UV-C devices, ranging from handheld wands to static, automated and conveyor systems, designed to provide surface/device disinfection, all the way to HVAC systems treating up to 10,000 scfm. Specific details are all protected by NDA, but lessons learned that can help move the UV industry forward are shared here. The recommended and employed evaluation approach involved the following steps: a) obtain desired specifications and objectives of the manufacturer; b) obtain all prior testing data and conclusions; c) develop an overall testing plan with the manufacturer, including any specific testing protocols requested (e.g., Modified ASTM E-2197); d) evaluate the device after set-up by the manufacturer to its specifications using calibrated twin radiometers (e.g., ILT-1700) and/or spectroradiometer (e.g., ILT-960) to produce a three-dimensional fluence rate (aka irradiance) map; and e) confirm UV performance and delivered fluence (dose) using biodosimetry runs with a minimum of two specified organisms (e.g. E. coli, MS-2 and/or T1UV bacteriophages).
The most surprising lesson learned was the general lack of understanding of basic UV light science and engineering factors. Developers often come from other fields and are more skilled in mechanical and electrical aspects, with the major focus on the UV-C design being to protect users from stray UV-C. Lacking, in most cases, is recognition that UV fluence (dose) distribution is critical and that devices are complex optical systems involving reflection, refraction and absorption of light by system and media (air or surface) components. A fundamental observation is that UV-C essentially is a line-of-sight technology that can provide developers with a basic understanding of some common design flaws. Further, URUVGI designs often fail to account for the UV-C fluence rate dropping at approximately the square of the distance from the UV-C source and the need for an unobstructed line-of-sight from source to target areas. Since UV-C sources are highly variable in terms of energy (wall-plug) efficiency, lamp type, wavelength(s) and life, clear and concise design specifications for the source are vital.
The UNH experience has been that a high percentage of the 30 devices evaluated did not achieve the manufacturer’s performance expectations without major modifications. Four primary reasons account for these outcomes:
- Poorly designed preliminary testing created unrealistic expectations. These often are performed on extremely small-scale bench-top proof-of-concept level (e.g., illuminating a static petri dish or 96 well plate with a UV-C source for hundreds of seconds to demonstrate that 99.9% inactivation could be achieved) and then extrapolating these results to the full-scale static or flow through equipment that often has not been fabricated yet.
- Preliminary testing performed with extremely poor scientific approach and/or QA/QC, such as lacking careful positive and negative controls, leading to erroneous conclusions (e.g., if system control losses are such that the UV-C device can only demonstrate 90% removal, it is not correct to claim that 10,000 test organisms were dosed initially and zero [ND] were detected after treatment; hence, the experiment proves 99.99% inactivation).
- Significant regions of the full-scale equipment where the multidirectional (these are not collimated beam systems) UV fluence rate (irradiance) measurements were at or below detection limits, especially as distance from the UV-C source increases (typically referred to as “cold spots” in the industry), indicating that regardless of the “long” contact times (e.g., 300 to 600 seconds) the resulting UV fluence (dose) essentially is zero in that region. These cold spot results are confirmed by biodosimetry results in each case.
- The specified inactivation is unrealistically high, does not entify target pathogens or neglects the impacts of interferences that absorb UV-C (e.g., dirt on surfaces). Further, for UV-C systems in a flowing HVAC duct or in URUVGI, the residence time distribution of air exposed to a given UV fluence rate is critical, highlighting the importance of understanding air mixing.
First Do No Harm
Potential Impacts on Critical Building Infrastructure
Integrating complex UV-C systems to treat HTS into critical building infrastructure has the potential to introduce new failure modes as well. The introduction of UV-C enclosure devices into a high-cycle environment (e.g., busy hospital elevators, restrooms, etc.) increases maintenance overhead and creates another single point of failure for these critical facility components. The closure of elevators or restrooms due to UV-C system failures would lead to a loss of owner and consumer confidence in the technology without improving the facility’s hygiene.
Misconceptions that Place the Multibarrier Approach at Risk
UV-C technology can improve healthcare facility hygiene, but it is never intended to replace the other well-established and cost-effective barriers, which include hand hygiene, “no-touch” technologies, air filtration and indoor ventilation rates (exchanges per hour). Far too many facility decision makers look at UV-C technologies as replacements to achieve cost savings.
Hand Hygiene (The “Gold Standard”): Numerous publications identify hand hygiene, normally achieved by placing a hand sanitizer dispenser at the critical points in healthcare facilities, as the most effective approach. 8, 9 Hands are the primary vector for pathogen transport. Disinfecting the skin surface addresses all subsequent touchpoints whether they are related to elevators or restroom HTS. 8, 9
AI-Based Touchless Technology Systems: To achieve a “no-touch” environment, software-based solutions (AI face and voice recognition) are used increasingly to improve hygiene in healthcare facilities. 10 These technologies represent prevention, reducing the need for HTS disinfection while providing positive user experiences.
Ventilation Rates and Air Filtration: ANSI/ASHRAE/ASHE Standards 62.1, 170 and 241 (2021-2025) provide solid guidelines (https://www.ashrae.org/technical-resources/standards-and-guidelines) for the use of ventilation rates, air filtration and the role of UV technologies to improve those systems in healthcare facilities. In all cases, the role of UV technologies (aka UVGI) is intended to augment and improve overall efficiencies, not replace ventilation and air filtration technologies (e.g., MERV 13 or higher).
References
- Thompson, Derek (2020). “Hygiene Theater Is a Huge Waste of Time” article in The Atlantic, https://www.theatlantic.com/ideas/archive/2020/07/scourge-hygiene-theater/614599/
- Balasubramanian, Chandana (2023). “What is the Chain of Infection and How to Break It” eBook GIDEON Informatics Inc., https://www.gideononline.com/blogs/what-is-the-chain-of-infection-how-to-break-it/
- V.C.C. Cheng, P.H. Chau c, W.M. Lee, S.K.Y. Ho, D.W.Y. Lee, S.Y.C. So, S.C.Y. Wong, J.W.M. Tai, K.Y. Yuen (2015) “Hand-touch contact assessment of high-touch and mutual-touch surfaces among healthcare workers, patients, and visitors” Journal of Hospital Infection Volume 90, Issue 3, Pages 220-225. https://doi.org/10.1016/j.jhin.2014.12.024
- American Conference of Governmental Industrial Hygienists (ACGIH) 2026Threshold Limit Values and Documentation: Ultraviolet Radiation. ACGIH; Cincinnati, OH, https://www.acgih.org/ultraviolet-radiation/
- Mphaphlele M, Dharmadhikari AS, Jensen PA, Rudnick SN, van Reenen TH, Pagano MA, Leuschner W, Sears TA, Milonova SP, van der Walt M, Stoltz AC, Weyer K, Nardell EA. (2015) “Institutional Tuberculosis Transmission. Controlled Trial of Upper Room Ultraviolet Air Disinfection: A Basis for New Dosing Guidelines.” Am J Respir Crit Care Med. 2015 Aug 15;192(4):477-84. doi: 10.1164/rccm.201501-0060OC. PMID: 25928547; PMCID: PMC4595666.
- Reed NG. (2010) “The history of ultraviolet germicidal irradiation for air disinfection.” Public Health Rep. 2010 Jan-Feb;125(1):15-27. doi: 10.1177/003335491012500105. PMID: 20402193; PMCID: PMC2789813.
- Wells WF, Wells MW, Wilder TS. (1942) “The environmental control of epidemic contagion I: an epidemiologic study of radiant disinfection of air in day schools.” Am J Hyg; 35:97-121.
- Nieradko-Iwanicka B. (2020) “Hand Hygiene – gold standard not only in prevention of COVID-19 infection.” Reumatologia ;58(4):191-195. doi: 10.5114/reum.2020.98429. Epub 2020 Aug 31. PMID: 32921824; PMCID: PMC7477480.
- Kasujja H, Waswa JP, Kiggundu R, Murungi M, Kwikiriza G, Bahatungire R, Kajumbula H, Alombah F, Joshi MP, Konduri N. (2024) “Enhancing infection prevention and control through hand hygiene compliance in six Ugandan hospitals using quality improvement approaches.” Front Public Health. Oct 22;12:1465439. doi: 10.3389/fpubh.2024.1465439. PMID: 39502813; PMCID: PMC11534609.
- Iqbal MZ, Campbell AG. (2021) “From luxury to necessity: Progress of touchless interaction technology.” Technol Soc. 2021 Nov; 67:101796. doi: 10.1016/j.techsoc.2021.101796. Oct 27. PMID: 36313277; PMCID: PMC9595506.

