By Richard A. Rasansky, CEO, and Benjamin D. Robertson, Ph.D., senior director of test method development and analytics, XCMR Inc.
Authors’ Note: This article provides a high-level summary of a two-year research and development project focused on advancing the state of the art in surgical site infection (SSI) control through non-pharmaceutical medical countermeasures. In alignment with a planned submission of a peer-reviewed academic research paper on this work, the detailed methods and supporting data are intentionally excluded from this article to maintain eligibility for journal publication. This overview is intended to share the scope and objectives of the effort, while comprehensive results and analyses will be presented in a forthcoming scholarly manuscript. Elements of this work were presented, including demonstrations and conceptual devices, at the 2025 IUVA World Congress in Lisbon.
Skin, soft tissue and surgical site infections (SSTI/SSI) remain among the most persistent and costly complications across healthcare systems worldwide. From outpatient and dental procedures to trauma care and military medicine, infections acquired at or near the time of tissue exposure contribute to patient morbidity, prolonged recovery, repeat interventions and escalating costs. 1 Despite decades of progress in sterile technique, antisepsis and antimicrobial therapy, SSI rates remain stubbornly high across many procedure types.
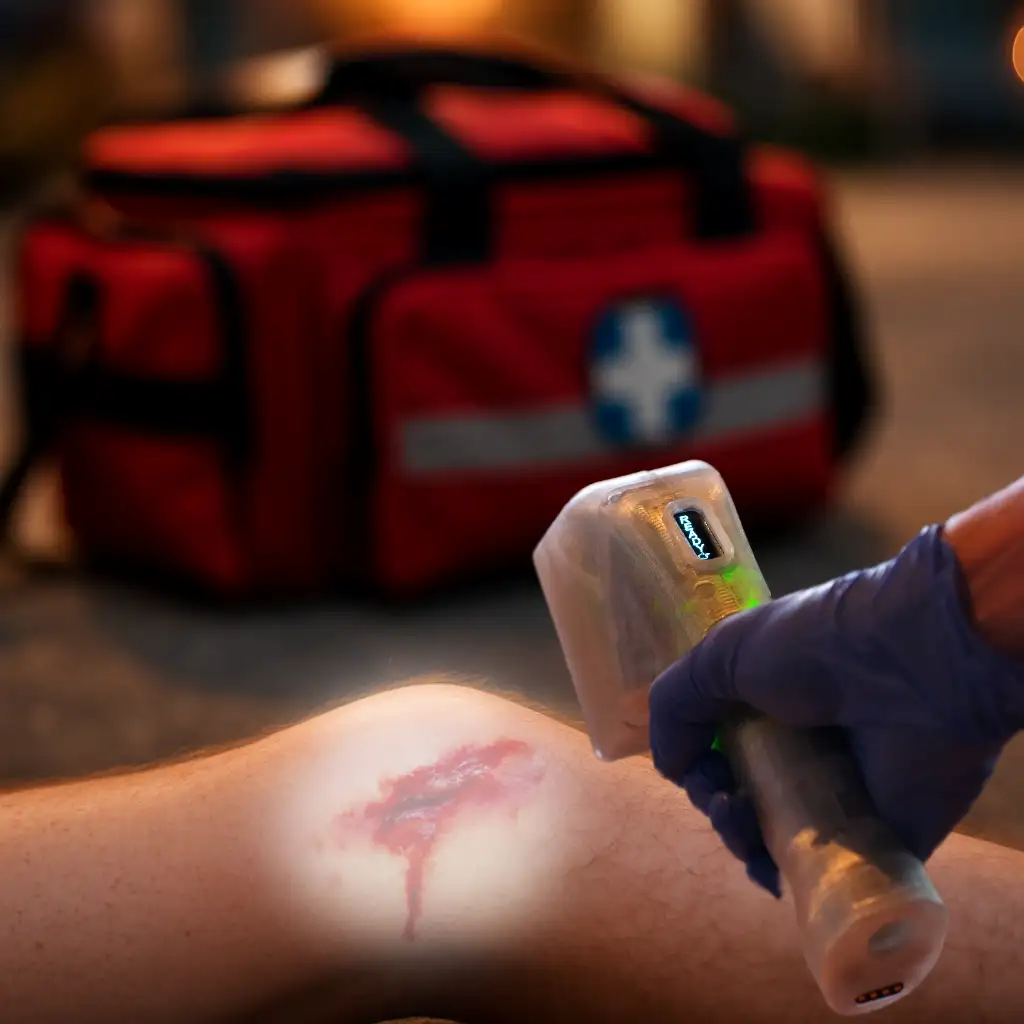
A central limitation of current infection-control measures is they are applied before or after a procedure, such as pre-operative skin preparation, post-operative antibiotics or wound dressings, leaving a critical gap during the procedure itself. Yet it is precisely during exposure, incision and manipulation of tissue that microbial contamination and inoculation risk are highest. 2 At present, there is no method to reduce microbial burden within the exposed surgical field during these moments of peak vulnerability.
Compounding this challenge is the accelerating crisis of antimicrobial resistance (AMR). As resistant organisms become more prevalent, reliance on systemic antibiotics alone becomes increasingly fragile. This reality has heightened interest in physics-based, pathogen-agnostic approaches that can reduce microbial burden in real time.
Against this backdrop, the authors’ company has led a multi-year research and development effort to explore whether photonic energy, applied safely and precisely, can serve as a new, procedure-integrated layer to reduce risk of SSI.
Photonic Inactivation: A Broad-Spectrum Mechanism for Disinfection
Ultraviolet (UV) irradiation has long been recognized for its ability to inactivate microorganisms by damaging nucleic acids and proteins essential for replication and function. UV-based approaches act through fundamental physical interactions shared by all known microorganisms, whereas antibiotics target specific biochemical pathways. Serial exposure to antibiotics has been shown to readily engender resistance. In contrast, serial passages of multidrug-resistant bacteria to UV did not engender resistance to UV and thus are unlikely to generate UV-resistant microbes when applied in the clinical setting. 3
Historically, UV disinfection has been used primarily for environmental and surface applications, such as air disinfection, water treatment and room or equipment decontamination, rather than for direct application to human tissues. This limitation has been driven by safety considerations, dose-control challenges and the absence of form factors suitable for point-of-care use.
Recent advances in emitter technology, dosimetry and control systems have begun to change that dynamic. Within this framework, the authors’ company demonstrated that a pulsed-xenon photonic platform designed for direct application to exposed tissue can achieve a greater reduction in local microbial bioburden that exceeds published data for topical antiseptics such as chlorhexidine. 4 Importantly, this enhanced antimicrobial performance was observed without visible evidence of harm to surrounding healthy tissue, supporting the feasibility of applying photonic disinfection directly at the wound or surgical site rather than limiting its use to environmental surfaces.
A Unified Research Program: From Source Selection to Practical Use
Over the past two years, XCMR has pursued a unified body of work spanning three tightly linked questions:
- Which photonic emitters are most effective at inactivating clinically relevant SSI pathogens under realistic constraints?
- Can those same emitters demonstrate meaningful reductions in microbial burden in biologically complex settings, while remaining within established safety limits?
- How can such technology be translated into a practical, clinician-usable tool that integrates into real procedural workflows?
Evaluating Emitter Classes for SSI Applications
A critical early step in this program was the comparative evaluation of multiple commercially available photonic emitter classes suitable for potential clinical applications. These included pulsed xenon flashlamps, krypton chloride (KrCl*) excimer sources operating in the Far-UV range and UV-C light-emitting diodes (LEDs) with narrow spectral output.
Each emitter class offers distinct advantages and tradeoffs with respect to spectral characteristics, intensity, exposure duration, manufacturability and alignment with established occupational exposure limits for human skin and eyes. Rather than optimizing for a single parameter, the approach emphasized system-level performance, balancing microbial inactivation efficacy with safety margins and practical deployment considerations.
The work demonstrated that not all UV-C sources perform equivalently when evaluated against common SSI pathogens under comparable constraints. Enhanced/broad-spectrum pulsed xenon sources consistently showed superior antimicrobial performance while enabling short exposure times compatible with procedural workflows. The pulsed-xenon approach demonstrated markedly higher local microbial reduction under comparable safety constraints. In contrast to conventional pre-operative chemical antisepsis, including chlorhexidine-based preparation, a shift toward physics-based, in-procedure prevention offers additional risk mitigation for SSI.
Importantly, these conclusions are not species-specific, as UV offers broad-spectrum effectiveness across gram-positive and gram-negative bacteria, including strains relevant to both civilian and military healthcare contexts.
Moving Beyond the Petri Dish: Biological Complexity Matters
While in vitro performance provides essential insight into emitter behavior, SSI risk arises in far more complex biological environments. Skin topography, tissue heterogeneity, fluids and host responses all influence microbial persistence and survival. Recognizing this, the authors’ company advanced selected emitter classes into pre-clinical evaluation designed to better approximate real-world conditions.
These studies demonstrated that photonic treatment applied prior to incision could meaningfully reduce viable microbial burden at the tissue site, with effects persisting beyond the immediate post-exposure period. Equally important, these reductions were achieved without visible evidence of collateral injury to surrounding healthy tissue. This combination of high local potency and tissue compatibility distinguishes photonic approaches from many existing antimicrobial strategies.
Why Timing Matters in SSI Prevention
Most SSI prevention strategies focus on reducing microbial burden before or after surgery. Yet contamination often occurs during exposure, when tissue is open, manipulated and temporarily deprived of normal barriers. Preoperative chemical antiseptics such as chlorhexidine provide important but transient surface protection. In contrast, photonic approaches can deliver substantially greater real-time microbial reduction within the surgical field or wound, addressing a long-standing vulnerability in SSI prevention and complementing existing standards of care.
Translating Science into a Usable Clinical Tool
Demonstrating biological effectiveness is only part of the challenge. For any SSI mitigation technology to have a real impact, it must integrate seamlessly into clinical workflows without adding complexity, time burden or new safety risks.
Guided by this principle, the development effort extended beyond emitter selection into human-factors engineering and usability design. Key considerations included ergonomic form factors suitable for constrained procedural environments; real-time user feedback to support consistent application; automated safety controls aligned with established exposure frameworks; and integrated video documentation capabilities to support traceability and future clinical integration.
While still under refinement, these systems illustrate how photonic energy could evolve from a laboratory concept into a practical, point-of-care tool to reduce microbial bioburden throughout the clinical procedure to help mitigate SSI. Crucially, the intent of such devices is not to replace antibiotics, antisepsis or sterile technique, but to serve as an additive layer, acting locally and in real time to reduce microbial burden at the site where infection begins.
Implications for Antimicrobial Resistance and System Resilience
The broader significance of this work extends beyond any single device or procedure. As AMR continues to erode the effectiveness of existing therapies, prevention-oriented strategies that reduce infection probability upstream become increasingly valuable.
By acting through fundamental physical mechanisms, photonic approaches offer several strategic advantages: pathogen-agnostic activity independent of resistance profiles; localized action that minimizes systemic exposure; and compatibility with existing standards of care rather than competition with them. In both civilian healthcare and military medicine, where contaminated wounds, delayed care and austere environments amplify infection risk, the ability to deploy portable, energy-based prevention tools could meaningfully enhance resilience.
Looking Ahead
The demonstration that a pulsed-xenon photonic platform can deliver order-of-magnitude greater local antimicrobial effect than conventional pre-surgical antisepsis, while remaining compatible with healthy tissue, suggests a significant opportunity to reimagine how and when antimicrobial protection is delivered during procedural care.
Ongoing efforts are focused on engineering refinement, procedural integration and generation of clinical and operational evidence to better define use cases, workflows and outcome impact. As healthcare systems confront rising infection burdens and diminishing antimicrobial options, the case for layered, prevention-first strategies continues to strengthen. Further clinical and translational studies are underway.
Acknowledgment and Disclaimer: This material is based upon work supported by the Air Force Research Laboratory, AFWERX and AFRL/RGKB under Contract No. FA864924P0393. Any opinions, findings and conclusions or recommendations expressed in this material are those of the author(s) and do not necessarily reflect the views of the Air Force Research Laboratory and AFWERX, AFRL/RGKB.
References
- Centers for Disease Control and Prevention (U.S.), “Antibiotic resistance threats in the United States, 2019,” Centers for Disease Control and Prevention (U.S.), Nov. 2019. doi: 10.15620/cdc:82532.
- C. D. Owens and K. Stoessel, “Surgical site infections: epidemiology, microbiology and prevention,” J. Hosp. Infect., vol. 70 Suppl 2, pp. 3–10, Nov. 2008, doi: 10.1016/S0195-6701(08)60017-1.
- H. Choi et al., “Can multidrug-resistant organisms become resistant to ultraviolet (UV) light following serial exposures? Characterization of post-UV genomic changes using whole-genome sequencing,” Infect. Control Hosp. Epidemiol., vol. 43, no. 1, pp. 72–78, Jan. 2022, doi: 10.1017/ice.2021.51.
- D. Adams, M. Quayum, T. Worthington, P. Lambert, and T. Elliott, “Evaluation of a 2% chlorhexidine gluconate in 70% isopropyl alcohol skin disinfectant,” J. Hosp. Infect., vol. 61, no. 4, pp. 287–290, Dec. 2005, doi: 10.1016/j.jhin.2005.05.015.
Ben Robertson has a bachelor of science degree in applied physics from Emory University and a Ph.D. from the joint department of biomedical engineering at UNC Chapel Hill and NC State University. He has 10 years of combined experience in pharmaceutical and medical device development, with a primary focus on pre-clinical research and development. Robertson is an avid cyclist, skier and dog lover currently based in Philadelphia, Pennsylvania.
Richard Rasansky is a founder and technology executive with more than two decades of experience leading scientists, engineers and technologists to build real-world platforms that improve health, safety and security at scale. Specializing in biosecurity, systems standards and dual-use technology, Rasansky is a graduate of The Wharton School and attended the University of Pennsylvania School of Engineering and Applied Science.

