By Amy Hedrick, CEO, Cleanbox Technology Inc., and Matthew Hardwick, Ph.D., CEO of ResInnova Clinical Diagnostics
High-level disinfection (HLD) is a regulatory necessity required for some semi-critical and critical medical devices, a process that is time-consuming, labor-intensive and highly specific to each device. Low-level disinfection (LLD) is intended for non-critical devices, such as blood pressure cuffs, stethoscopes or certain wearable sensors. These LLD methods are (relatively) faster than HLD but require disinfection application and process precision. The efficacy of this process is highly dependent on proper application technique, which can vary widely in real-world settings. Surface coverage, contact time and human error all influence disinfection outcomes. Studies have shown that compliance with LLD protocols is inconsistent, particularly in fast-paced clinical environments where equipment turnover is high and staff are multitasking. 1
Many reusable medical devices now incorporate complex geometries, touchscreen interfaces and multi-material assemblies that are difficult to clean and disinfect thoroughly. Traditional LLD protocol may not penetrate crevices or effectively sanitize soft-touch plastics, fabrics or internal surfaces, and the liquid, chemicals and heat components of LLD can cause device degradation and failure.
This gap between protocol and practice raises the important question: How can we increase effective and practical disinfection compliance without increasing complexity at the point of care?
These challenges underscore the need for adjunct or alternative methods, such as GUV, that can provide reliable, validated disinfection with less dependence on operator performance and surface accessibility, especially in high-risk or high-throughput areas like emergency departments, diagnostic labs and long-term care. Interestingly, after a lull in participation following 2022, GUV manufacturers made a resurgence at this year’s annual conference for the Association for Professionals in Infection Control and Epidemiology (APIC). 2 This upturn in GUV participation signals a re-establishment of both interest in and utility of GUV technologies in healthcare for infection prevention.
Using GUV to Address the Challenges of Traditional Disinfection Methods
Unlike chemical disinfection, GUV systems offer rapid, non-liquid disinfection through the targeted application of UV-C light. Properly designed enclosure-based GUV devices can achieve consistent, validated reductions in microbial load within minutes – without soaking, rinsing or chemical residues. This makes the process faster, more scalable and often more compatible with sensitive device materials.
Importantly, GUV can simplify workflows in settings where multiple devices need to be disinfected frequently and reliably. By minimizing chemical exposure and operator variability, GUV technology reduces the margin for error while increasing throughput. Still, widespread adoption requires more than efficiency; it depends on robust testing protocols, performance validation and regulatory recognition.
As interest in GUV grows, so does the need for formalized guidance. Regulatory bodies will require consensus on test methods, efficacy endpoints and appropriate labeling. But the potential upside is significant: a standardized, validated and scalable method of disinfection that bridges the gap between what is required and what is operationally achievable on the front lines of care.
Addressing Gaps in Disinfection and Sanitization Protocol
Disinfection of modern medical devices is extremely difficult. There is a vast array of devices used in patient care, ranging from traditional devices, such as blood pressure cuffs and blood oximeters, to non-traditional devices, such as smartphones and electronic tablets. Cleaning methods must be compatible with each of these devices. Saturation of a device with a liquid disinfectant may be effective on some devices, but it certainly will not work with other devices, such as those with delicate electronic interfaces.
Disinfection methods must be compatible with an incredible variety of different materials used to make medical devices. Harsh disinfectants may not be appropriate for repeated use with a sensitive touchpad or other types of plastics. Another consideration is the geometries of certain medical devices. For example, devices with ridges are difficult to clean with a wipe, as are devices with crevices.
There are additional risk factors associated with relying on manual sanitation, including constraints of chemical applications on sensitive materials, a broad and inconsistent range of application efficacy, and minimal compliance assurance.
GUV offers a unique solution to cleaning modern medical devices. While not a silver bullet, well-designed GUV devices may be used in concert with less harsh liquid cleaners and disinfectants to achieve results equal to those of standard disinfectants.
- Saturation. There is no requirement for saturation of a device with any liquid disinfectant, and this is significant in the life span of the device being disinfected, as well as the time required to complete disinfection (no leave-wet time as with all other liquids and chemicals). GUV requires the removal of visible soil in order for its light to reach the surface to be cleaned. Therefore, the removal of soil may be achieved with a gentle cleanser and physical abrasion.
- Material Compatibility. Lower GUV doses, especially those with narrow-spectrum light and those utilizing form factors such as LED, pose little risk to the integrity of most materials even with repeated uses, with independent data supporting the premise that rapid dosage at a specified exposure does not cause impactful material degradation, even with frequent repetition over time.
- Geometries. GUV is light. Therefore, for all practical purposes, it travels in a straight line. However, with the incorporation of movement and broad surface coverage engineering of either the GUV light source or the medical device, it is possible to cover practically all ridge geometries and crevices.
Benefits of Testing with Carriers
Given the complexities of modern medical devices, it is critical that each GUV device is tested for its antimicrobial efficacy on the medical devices to be disinfected. If one considers all the possible antimicrobial testing that must be performed for each GUV device, such as use with different cleaners, against various microbes (bacterial, fungal and viral), and the vast array of devices on the market, proper antimicrobial efficacy testing on every possible combination is practically impossible from both a financial and time standpoint. Therefore, a more streamlined approach is called for during the initial validation stages for each GUV device.
While medical devices are composed of numerous material types, most materials fall into just a few categories: hard or soft plastics, porous or non-porous textiles, metal and glass. Rather than test all medical device materials, it is possible to test against key representatives of each category. Additionally, rather than test against each fully composed medical device, testing against the materials used in these devices allows for early validation.
It is possible to create coupons or carriers for each of these material types. With smaller, easy-to-handle carriers, a significant number of variables may be tested in a relatively short period of time and at a much lower price point. While it is critical for performance of final validations on actual medical devices, such as for an FDA regulatory submission, carriers are an expedient and relatively high throughput and utilitarian option as a first step validation.
Medical Device GUV Protocol: Developing Testing Standardization
Historically, determining the antimicrobial efficacy of GUV devices on surfaces has been left up to the testing group (air and water GUV testing is more established). This lack of a common standardized test methodology has led to a great number of studies on GUV surface antimicrobial efficacy that do not always reach the same conclusions. 3,4 During the last few years, however, efforts have been made by various international standards organizations to fill in this gap. Below is a list of current GUV standards (practices, guides, test methods and performance) from international standards organizations. This list is not intended to be comprehensive as there are several regional standards like those listed below.
- ASTM E3135-25 Standard Practice for Determining Antimicrobial Efficacy of Ultraviolet Germicidal Irradiation Against Microorganisms on Carriers with Simulated Soil. This standard practice is meant to provide a foundation for future standard test methods. It defines test conditions, including organic soils, for evaluating the antimicrobial efficacy of GUV device surfaces.
- ASTM E3179-25 Standard Test Method for Determining Antimicrobial Efficacy of Ultraviolet Germicidal Irradiation against Influenza Virus on Fabric Carriers with Simulated Soil. As the name indicates, this standard test method evaluates the antiviral efficacy of GUV devices against Influenza A on textile carriers.
- ANSI/ASHRAE Standard 185.4-2024 Method of Testing In-Room Ultraviolet Devices and Systems for Microbial Inactivation on Surfaces in a Test Room. This standard test method was designed to specifically determine the antimicrobial efficacy for whole-room GUV devices. It is agnostic of the source of the GUV and of the device design (stationary or automated movement).
- BS 8628:2022 Disinfection using ultraviolet radiation. This British quantitative standard test method assesses direct illumination GUV antimicrobial efficacy. This method also specifies performance criteria for each microbial classification.
- ANSI-HSI 2000-2023 Healthcare UV Germicidal Light Whole-Room Disinfection Systems. This performance standard establishes antimicrobial efficacy requirements for whole-room GUV devices in both patient and operating rooms. Unlike other GUV standards, ANSI-HSI 2000-2023 is geared towards providing healthcare professionals charged with infection prevention the data they need to compare GUV devices across all platforms.
It should be noted that each of these standards utilizes carriers of various materials to provide consistency and ease of use for both large- and small-scale testing. Also of note, several of these standards have been born out of the International Ultraviolet Association’s (IUVA’s) Healthcare/HAI Working Group. At a recent joint conference between the IUVA and the National Institute of Standards and Technology (NIST), titled “Advancing UV Germicidal Efficacy Measurement: Metrology, Standards, Data and Protocols,” a commitment was made to develop a core antimicrobial efficacy standard test methodology. This method will provide a level-setting series of testing procedures designed to ensure that all laboratories worldwide are working with microbes and reagents identical in their response to GUV.
Testing Design for Commercial GUV Product
The following study was built to test out GUV efficacy on medical devices using a proprietary enclosed GUV-LED-based product (Cleanbox® Omniclean®).The purpose of this study of GUV on a resin-based, semi-porous material probe bracket (Mediview® proprietary device) was to determine the antimicrobial efficacy of the Cleanbox OmniClean narrow-spectrum GUV device against common pathogens and to develop a GUV-based cleaning method for a range of medical devices.
Note: This data was first published as part of “EM Probe Bracket Cleaning Method Development Utilizing Germicidal Ultraviolet Light (GUV)” in KILMER Conference 2025: Next Generation Microbiological Quality & Sterility Assurance. McDonnell (ed). In press. A. Hedrick, M. Hardwick, TJ Woody, G. Conrado (2025).
Study
Purpose of Study
- Evaluate the antimicrobial efficacy of the Cleanbox OmniClean narrow-spectrum germicidal ultraviolet (GUV) device on a resin-based, semi-porous medical probe bracket (Mediview proprietary device).
- Develop a GUV-based cleaning protocol for various medical devices.
Methods
- Antimicrobial Efficacy Testing:
- Magnetic, stainless-steel carriers were inoculated with either 6.39-log colony-forming units (CFU) Methicillin-resistant Staphylococcus aureus (MRSA; ATCC 33591) or 5.09-log foci-forming units (FFU) H1N1 Influenza (ATCC VR-1469).
- Stainless steel carriers were attached to items (eyeglasses, mobile phone, stethoscope diaphragm/earpiece) and exposed to different GUV doses. Dose was not directly measured, but was based off of device run time.
- Following recovery into recovery buffer, log reduction of each microorganism was measured relative to unexposed (negative) controls.
- Cleaning Protocol Development:
- A representative medical device was inoculated with five representative organisms.
- Devices were exposed to varying GUV doses, and log reductions relative to unexposed (negative) controls were recorded. Dose was not directly measured, but was based off of device run time.
Results
- H1N1 Influenza Virus:
- Devices inoculated with 5.09-log viral particles.
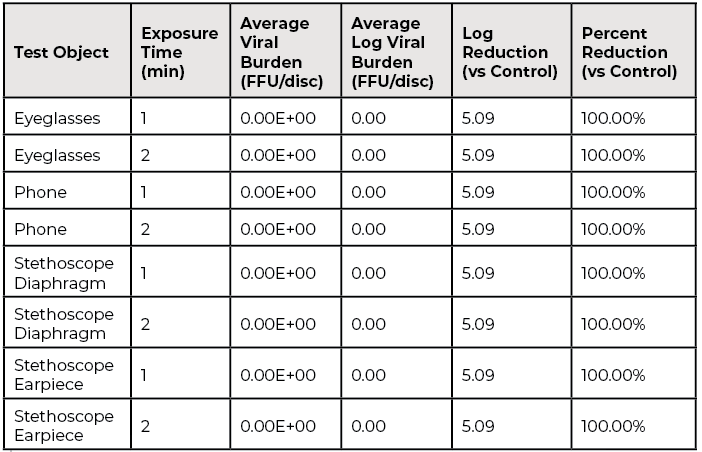
- After either one or two minutes of GUV exposure (carriers were exposed to two different run times), 100% inactivation (5.09-log reduction) was achieved on all tested surfaces (eyeglasses, phone, stethoscope diaphragm/earpiece).
- MRSA:
- Devices inoculated with 6.33-log MRSA CFU.
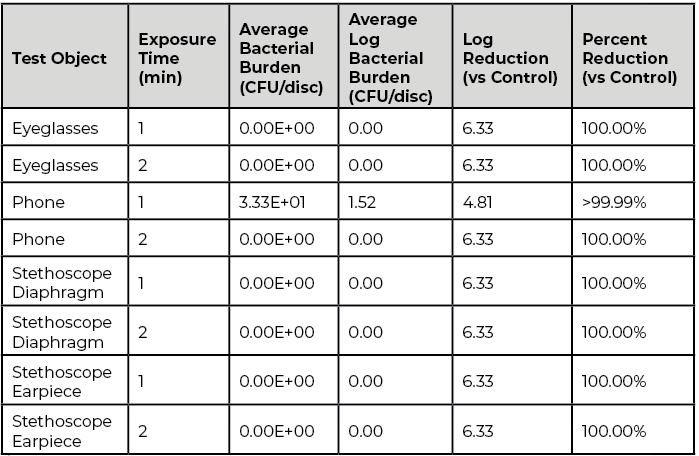
- After one or two minutes of GUV exposure, eyeglasses, stethoscope diaphragm, earpiece: 100% reduction (6.33-log reduction); mobile phone: >99.99% reduction (4.81-log) at 1 minute; 100% reduction at 2 minutes.
Results
OmniClean GUV Inactivation of H1N1 Influenza Virus: Stainless steel carriers were inoculated with 5.09-log of viral particles and attached to either eyeglasses, a mobile phone or a stethoscope diaphragm. Carriers then were exposed to two different run times, either one or two minutes, in the OmniClean GUV enclosure. For each test object and exposure time, no viral particles were recovered (5.09-log reduction; see Table 1).
OmniClean GUV Reduction of Methicillin-resistant Staphylococcus aureus (MRSA): Stainless steel carriers were inoculated with 6.33-log MRSA colony forming units (CFU) and attached to either eyeglasses, a mobile phone or a stethoscope diaphragm. Carriers were then exposed to either one or two minutes in the OmniClean GUV enclosure. For each test object and exposure time, greater than 4-log reductions were achieved (see Table 2). In fact, all but one time point on the mobile phone resulted in 100% MRSA kill (6.33-log reduction).
Conclusion
This study is one of many steps in advancing the conversation around standardization in germicidal ultraviolet (GUV) disinfection for medical equipment. Data strongly supports the efficacy and utilitarian benefits of GUV and the increased outcomes with GUV as a highly effective adjunct tool for existing infection control protocols, successfully decreasing time and risk while addressing the need for an easy-to-implement process.
As healthcare environments seek scalable, repeatable solutions for infection control, GUV offers a promising adjunct to existing disinfection protocols, helping bridge the gap between real-world practicality and regulatory expectations.
Data: Matthew Hardwick, Cleanbox Technology, Azzur Laboratories (TJ Woody, Sr. Director, Cleaning Validation and Gabriella Conrado) and MediView XR. Study: Hedrick A, M Hardwick, TJ Woody, G Conrado (2025). EM Probe Bracket Cleaning Method Development Utilizing Germicidal Ultraviolet Light (GUV). In, KILMER Conference 2025: Next Generation Microbiological Quality & Sterility Assurance. McDonnell (ed). In press.
Laboratory: Azzur Labs, ResInnova Laboratories
Researchers and Study: Cleanbox Technology (Amy Hedrick), Azzur Laboratories (TJ Woody, Sr. Director, Cleaning Validation and Gabriella Conrado), MediView XR. (Compliance team); Independent microbiologist (Dr. Matthew Hardwick).
Additional supporting data is available but not published.
References
- Carling PC, Bartley JM. Evaluating hygienic cleaning in health care settings: what you do not know can harm your patients. Am J Infect Control. 2010; 38: S41–50. https://doi.org/10.1016/j.ajic.2010.03.004 PMID: 20569855
- https://airmidhealthgroup.com/apic-2025-ultraviolet-comebacks-air-purification-momentum-and-the-certification-gap.html
- Boyce JM, Donskey CJ. Understanding ultraviolet light surface decontamination in hospital rooms: A primer. Infect Control Hosp Epidemiol. 2019 Sep;40(9):1030-1035. doi: 10.1017/ice.2019.161. Epub 2019 Jun 18. PMID: 31210119.
- Masjoudi M, Mohseni M, Bolton JR (2021) Sensitivity of Bacteria, Protozoa, Viruses, and Other Microorganisms to Ultraviolet Radiation. J Res Natl Inst Stan 126:126021. https://doi.org/10.6028/jres.126.021
